Valbenazine Tosylate
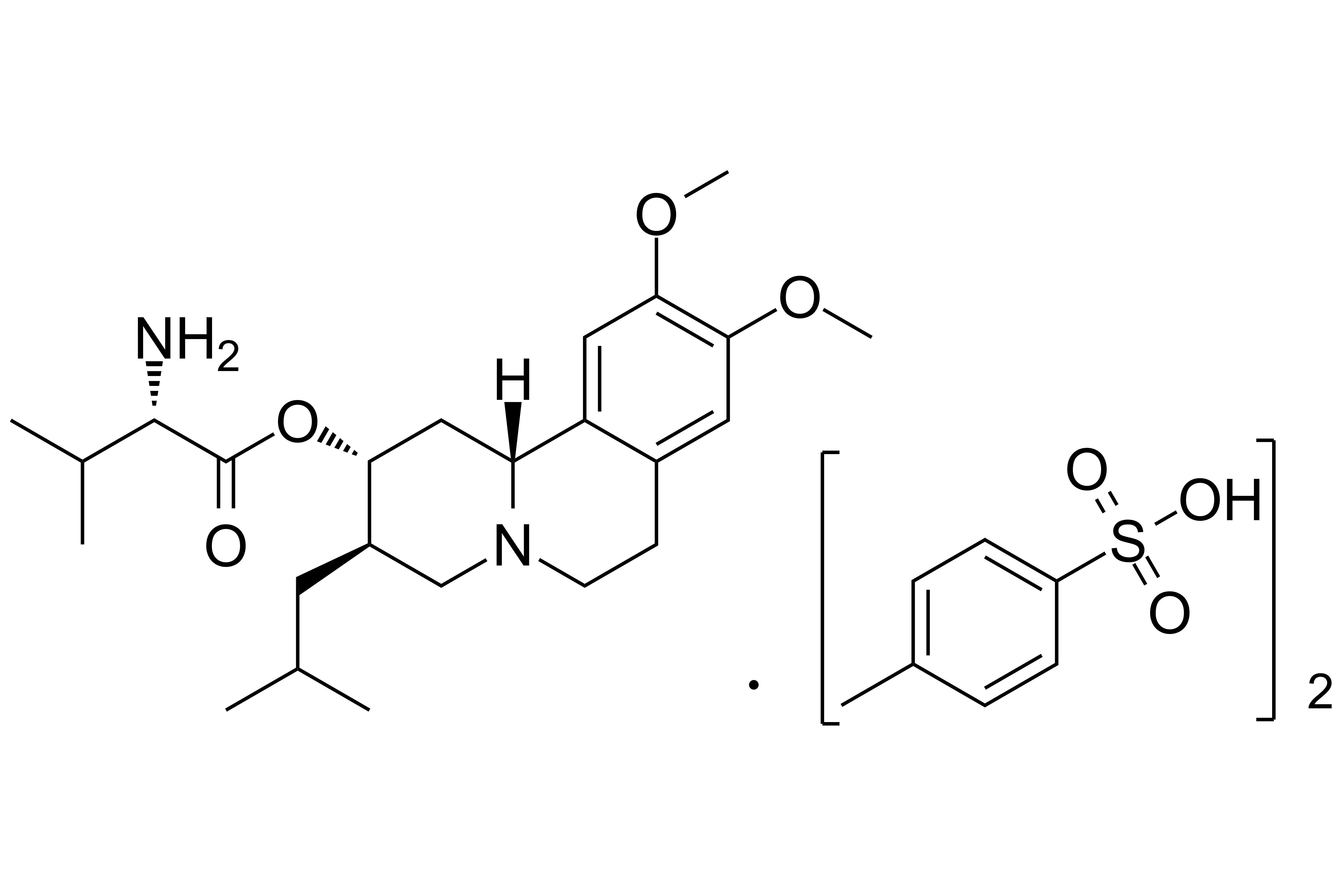
Valbenazine Tosylate
【Chemical Name】Valbenazine Tosylate
【Original】Neurocrine Biosciences
【Time to market】2017.4.11
【Patents expire】2029年10月6日
【Use】Tardive Dyskinesia
Valbenazine Tosylate
一、Product Overview
Valbenazine, developed by Neurocrine Biosciences for the treatment of adult tardive dyskinesia, was approved by the Food and Drug Administration (FDA) on April 11, 2017 and its patent expires in 2029.
二、Main Product
Description | Structural Formula | CAS No. | Category |
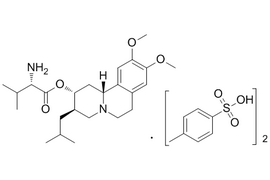 | 1639208-54-0 | API | |
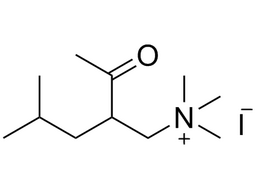 | 1069-62-1 | Intermediate | |
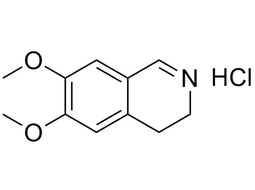 | 20232-39-7 | Intermediate | |
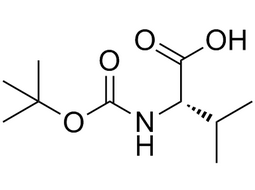 | 13734-41-3 | Intermediate |
Note: Products protected by valid patents are not offered for sale in countries where the sale of such products constitutes patent infringement and its liability is at buyer's risk.


