Suvorexant
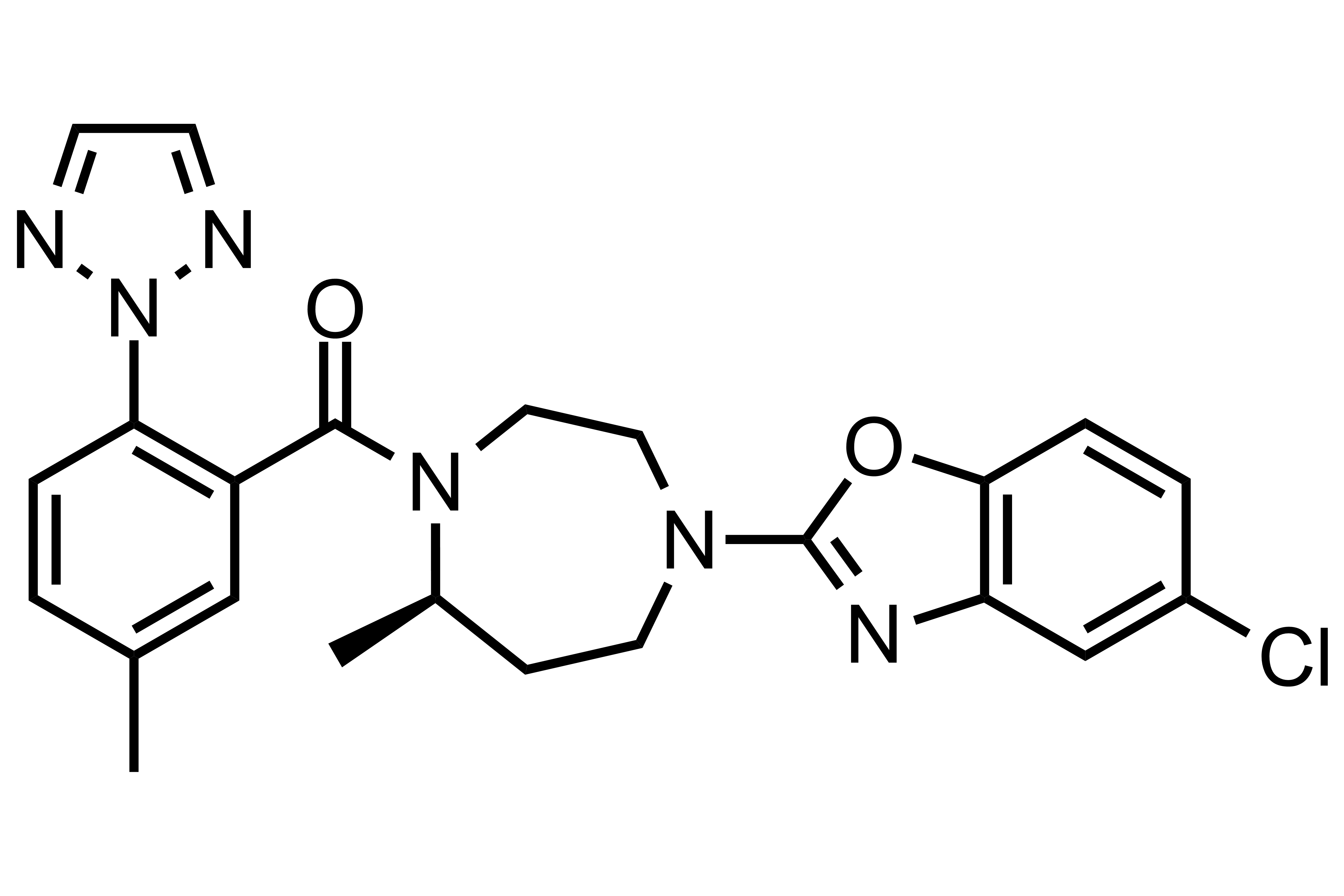
Suvorexant
【Chemical Name】Suvorexant
【Original】MSD
【Time to market】2014.8.13
【Patents expire】2027.11.30
【Use】Oral tablets are used to treat insomnia.
Suvorexant
一、Product Overview
Suvorexant was developed by Merk Sharp & Dohme (MSD) to treat insomnia. It was approved by the US Poetry and Drug Administration (FDA) on August 13, 2014. Then it was approved by the Japanese Medical Devices Comprehensive Agency (PMDA) on September 26, 2014. The compound patent expired on November 30, 2027.
二、Product Overview
Description | Structural Formula | CAS No. | Category |
Suvorexant (MK-4305) | 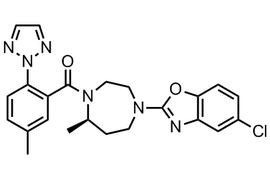 | 1030377-33-3 | API |
5-methyl-2-(2H-1,2,3-triazol-2-yl)benzoic acid | 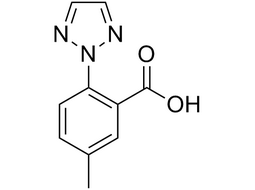 | 956317-36-5 | intermediates |
(R)-1-(4-Methoxy-benzyl)-5-methyl-[1,4]diazepane | 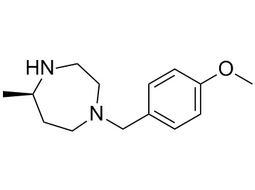 | NA | intermediates |
2,5-Dichlorobenzooxazole | 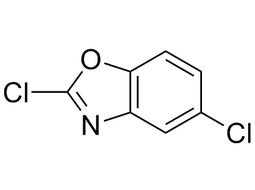 | 3621-81-6 | intermediates |
(R)-5-chloro-2-(5-methyl-1,4-diazepan-1-yl)benzo[d]oxazole | 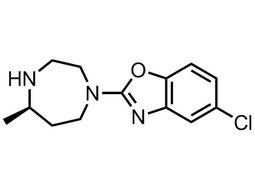 | 1266975-27-2 | intermediates |
Note: Products protected by valid patents are not offered for sale in countries where the sale of such products constitutes patent infringement and its liability is at buyer's risk.


