Vonoprazan Fumurate
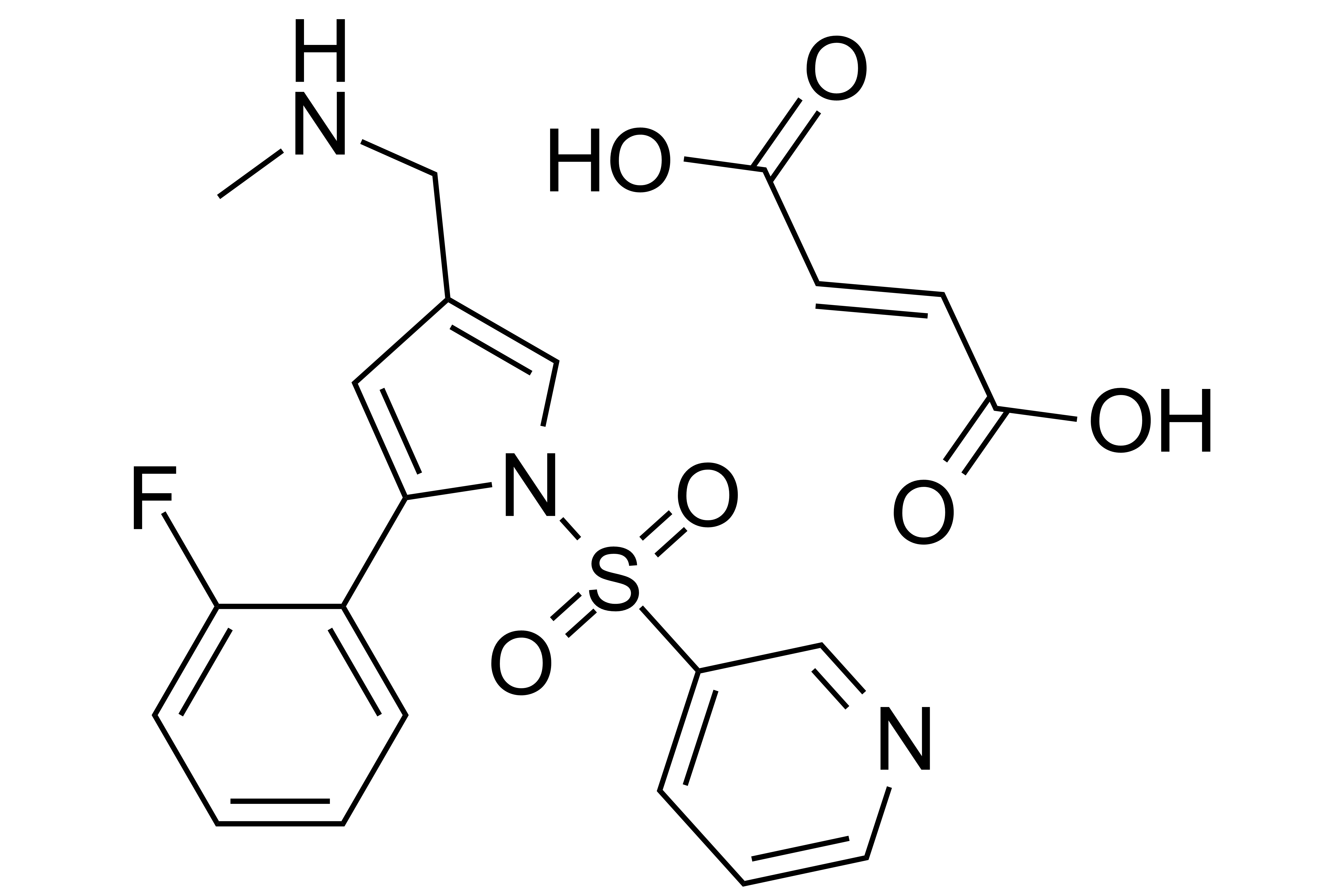
Vonoprazan Fumarate
【Chemical Name】Vonoprazan Fumarate
【Original】Takeda & Otsuka
【Time to market】2014.12.26
【Patents expire】2026.8.29
【Dosage and Usage 】Tablets: 10 mg and 20 mg of vonoprazan,used for healing of all grades of erosive esophagitis and relief of heartburn associated with erosive esophagitis in adults and to maintain healing of all grades of erosive esophagitis and relief of heartburn associated with erosive esophagitis in adults.
Vonoprazan Fumarate
一、Product Overview
Vonoprazan Fumarate was jointly developed by Takeda and Otsuka for the treatment of gastric ulcer, duodenal ulcer and reflux esophagitis. On December 26, 2014, Vonoprazan Fumarate was approved for market by PMDA, and the compound patent expired on August 29, 2026.
二、Main products
Description | Structural Formula | CAS No. | Category |
Vonoprazan Fumarate |  | 1260141-27-2 | API |
3-Pyridinesulfonylchloride | 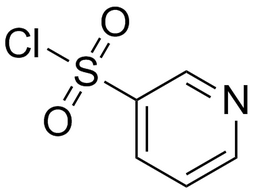 | 16133-25-8 | intermediates |
5-(2-fluorophenyl)-1H-pyrrole-3-carbaldehyde | 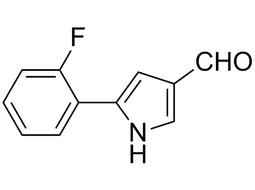 | 881674-56-2 | intermediates |
5-(2-fluorophenyl)-1-(3-pyridinylsulfonyl)-1H-pyrrole-3-carboxaldehyde | 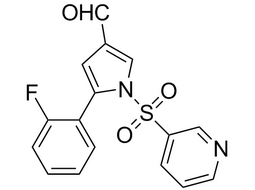 | 881677-11-8 | intermediates |
Note: Products protected by valid patents are not offered for sale in countries where the sale of such products constitutes patent infringement and its liability is at buyer's risk.


